Every year, thousands of therapeutic platelet units reach the end of their short 5-day shelf life and are discarded. Even though they are no longer usable for patients, they remain biochemically rich. More than 50–60% of prepared platelets worldwide go unused for transfusion, contributing to biomedical waste. But what if these expired platelets could be given a second life? One that advances science, reduces waste, and supports ethical innovation?
That’s precisely what recent evidence now supports: expired or outdated therapeutic-grade platelet concentrates are a safe, effective, and sustainable raw material for producing human platelet lysate (hPL), an effective alternative to fetal bovine serum (FBS) in cell culture.
Evidence Hierarchy: From case studies to systematic reviews
Our review of 17 global studies demonstrates a clear scientific consensus for repurposing expired platelets. The findings form a robust hierarchy of evidence:
- Systematic Reviews (n = 2): Aggregated data from multiple studies show no loss of safety or efficacy when expired platelets are used to make hPL.
- Literature Reviews (n = 2): Analyses confirm global acceptance of outdated platelet use and encourage moral responsibility to reduce waste.
- Comparative Laboratory Studies (n = 10): Experimental evidence confirms that expired platelet lysates perform equivalently to those from fresh platelets.
- Case Reports and Opinion Papers (n = 3): Expert consensus recognizes the ethical and technical value of reusing outdated platelet units.
Together, these form a solid scientific foundation for regulatory acceptance and industrial-scale implementation.
What the Studies Show
- Cell growth and differentiation remain unaffected: hPL from expired platelets successfully supports the growth of human mesenchymal stromal cells (hMSC), chondroprogenitors, and stem cells from bone marrow, adipose tissue, and dental pulp.
- Bioactivity remains potent after months of storage: Platelet concentrates stored for up to four (4) months at –80°C retain their cell-supportive growth factor profile.
- Platelet quality matters more than shelf time: Variations in platelet count and preparation technique influence consistency more than days post-expiry.
- No interference with patient supply: Repurposing expired platelets diverts non-transfusion material to research, removing competition for clinical-grade units.
Key Examples
- Jonsdottir-Buch et al. (2013): hPL from outdated platelets supported mesenchymal stem cell proliferation and osteogenic differentiation as effectively as fresh units (PLoS ONE).
- Glovinski et al. (2016): Outdated buffy coat samples are an efficient source for hPL to support adipose-derived stem cell expansion (Cytotherapy).
- Shanbhag et al. (2020): Demonstrated safe hPL production from platelets stored ≤4 months, with no difference in cytokine content or cell performance (Stem Cell Research & Therapy).
- Lee et al. (2023): Developed optimized freeze–thaw protocols to “up-cycle” expired platelet concentrate into a standardized, growth-factor–rich supplement (J. Clin. Med.).
A Circular Future for Blood Products
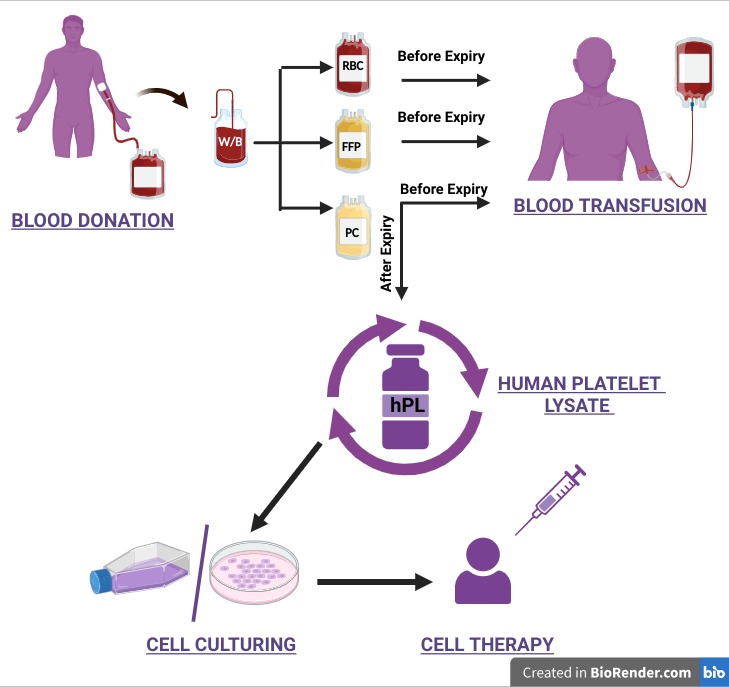
Repurposing expired platelets bridges an ethical and scientific gap. It aligns with GMP, ISBT, and WHO recommendations for the efficient use of human biological resources, while advancing the global transition to animal component–free biomanufacturing.
By adopting expired platelet lysate production, blood establishments can:
- Reduce biomedical waste
- Lower reliance on animal-derived reagents
- Support the cell and gene therapy industries
- Extend the value chain of blood donation programs
Conclusion
By embracing scientifically validated reuse through human platelet lysate production, we strengthen both biomedical innovation and sustainability — honoring every blood donation and every drop of potential it represents.

Leave a comment