Data collected between 25 March 2025 to 03 October 2025
Executive Summary
This report analyses survey data from 65 professionals across academia and industry regarding their use of Foetal Bovine Serum (FBS) in cell culture. The data revealed a near-universal reliance on FBS, though a significant majority of respondents report experiencing issues with its use. The primary driver for adopting any FBS alternative is equivalent performance, specifically regarding cell viability, morphology, and the ability to support cells across multiple passages.
1. Participant Demographics
The survey captured insights from 65 professionals, with a strong lean toward commercial applications while still maintaining an academic perspective:
- Total Respondents: 65
- Sector split:
- Academia: 32.3% (21 respondents)
- Industry: 67.6% (44 respondents)
2. Current FBS Usage and Pain Points
- Current Usage: 95% (62 out of 65) of the respondents are currently using FBS in their workflows. Applications range from basic research and stem cell culture to viral vector production and vaccine development.
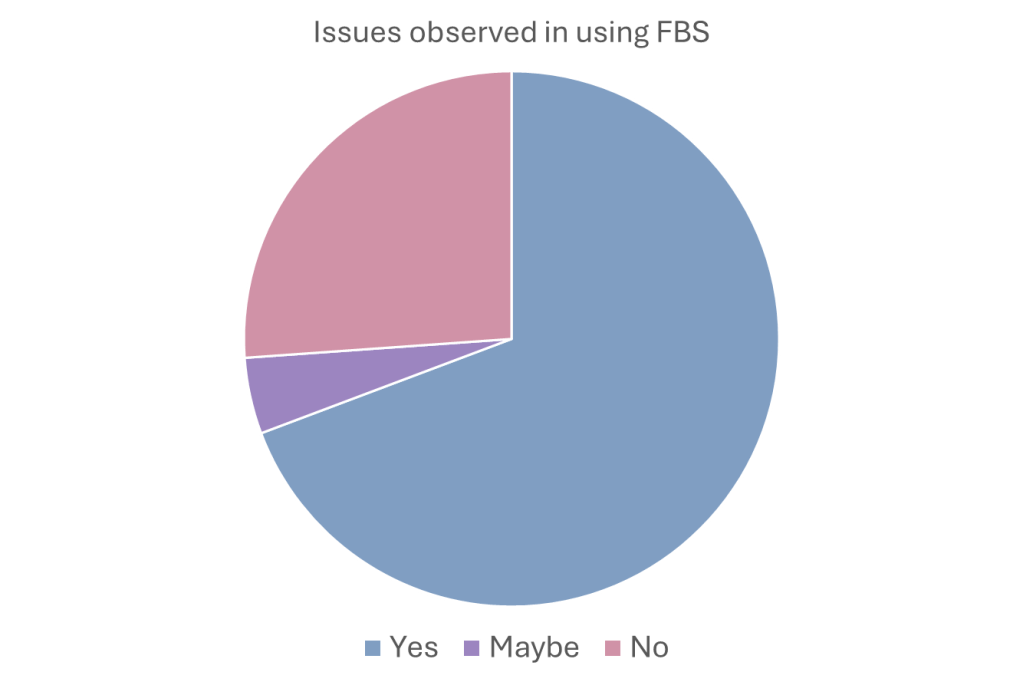
- Issues with FBS: When asked if they experienced issues using FBS, the responses indicated most users tolerated issues using FBS (73.8%), indicating low awareness/acceptance levels for alternatives.
- Qualitative Pain Points:
- Cell culture reproducibility concerns: 13.8% (9 out of 65) mentioned concerns regarding differences in cell viability across FBS of different geographical origins.
- Batch-to-Batch Variability: 9.2% (6 out of 65) mentioned inconsistency in between batches.
- Contamination Risks: 7.7% (5 out of 65) mentioned concerns regarding endotoxins, mycoplasma, and adventitious agents.
- Other concerns: animal-cruelty was only mentioned by one respondent.
3. Key Requirements for an FBS alternative
When asked what is most important in an FBS alternative, respondents clearly prioritized biological efficacy.
- Equivalent Performance to FBS (Top Priority): 50.8% (33 out of 65) of the respondents emphasized that alternatives must support proper cell growth, maintain healthy morphology, and allow for long-term passaging (specifically noting benchmarks like 10 to 15+ passages).
- Flexibility & Reproducibility: 23.1% (15 out of 65) of the respondents mentioned the need for alternatives that work across various cell lines and assays (e.g., HEK293, stem cells, viral assays) while providing consistent, reproducible experimental results.
- Quality & Consistency: 15.4% (10 out of 65) of the respondents mentioned the need for reliability in the product composition and low batch variability.
- Chemically-defined media: 7.7% (5 out of 65) desire for fully-defined media with optimal, known concentrations of growth factors to eliminate the “black box” nature of FBS.
- Regulatory Acceptance & Cost: Industry respondents heavily emphasized the need for regulatory compliance (especially for vaccine and viral vector production) and economic viability.
- Other: Only 1.5% (1 out of 65) of the respondents specifically highlighted “no animal cruelty involved” as a primary goal.

4. Limitations of the Data
Sample Size and Sector Skew: While the sample size of 65 provides solid directional data, the responses are heavily weighted toward industry professionals (67.6%). Therefore, the findings may underrepresent the specific pain points of academic researchers, such as publication pressures or grant-related limitations.
Low Baseline Awareness of Alternatives: The data suggests that users currently tolerate FBS issues due to a lack of awareness regarding viable alternatives. Consequently, respondents’ criteria for an alternative may be limited by their inability to envision a product that outperforms traditional FBS, rather than just matching it.
Open-Ended Response Bias: Because qualitative pain points were gathered via open-ended responses, the low mention of factors like cost (< 10 respondents), human-relevance (one respondent), and animal cruelty (one respondent) indicates these are not top-of-mind issues, but it does not necessarily mean respondents are entirely indifferent to them.
5. Recommendation and Next Steps
The data shows that users currently “tolerate” FBS. To drive adoption, ULQA Scientific will focus on market education to disrupt the Status Quo. By focussing on providing free samples, robust transition protocols, and develop case studies across various cell lines will help lower the perceived risk of switching, while addressing the notable reproducibility gap observed across users.
- Participate in the Survey or Request a free sample
- Get in touch to become a collaborator: FBS equivalence studies

Leave a comment